New Research Suggests Optimism for HIV/AIDS
For a scientist who studies the spread of HIV/AIDS — one of the worst pandemics on record — Brian Williams is surprisingly optimistic. To date, AIDS, the immunodeficiency syndrome caused by the virus HIV, has killed more than 35 million people worldwide. But Williams, co-founder of the South African Center for Epidemiological Modelling and Analysis (SACEMA), thinks we are close to effectively wiping out AIDS in eastern and southern Africa. This is despite the fact that there are more people living with HIV there than anywhere else in the world.
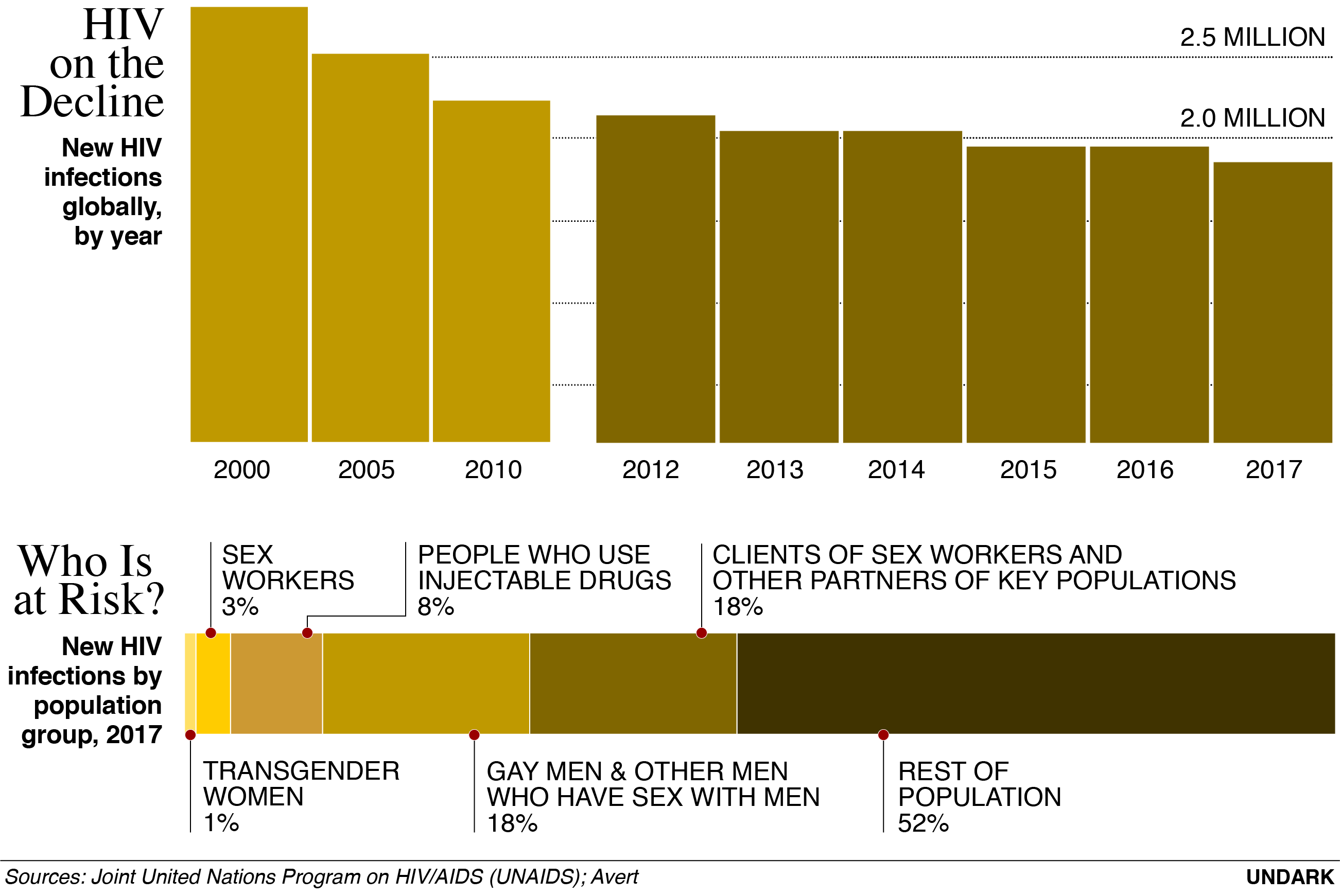
In August, Williams and Reuben Granich, an independent public health consultant who has worked on HIV control for more than two decades, published preliminary work that projects the continuing decline of HIV cases in many African countries over the coming years. By 2030, if trends in the use of HIV/AIDS treatments hold, the rate of infection might fall to a key threshold of one in 1,000 people in some of the worst-affected nations. That threshold would, in theory, stop the disease’s spread, Williams says.
“We can certainly end AIDS,” he adds. “It is a perfectly controllable disease, let me say.”
That’s a bold statement, and it’s based on the projections of computer models that look at the interplay between HIV infections and treatments. The models give researchers a target to hit, which is “a great thing to do because it causes us to think, ‘How do we reduce infections?’” says Anna Bershteyn, a senior research manager at the Institute for Disease Modeling. But Bershteyn and other experts are not convinced a one-in-1000 threshold means the epidemic will fade away.
That’s the thing about models: Each offers a slightly different view of the world and none can perfectly predict the future. But for HIV/AIDS, two factors may prove Williams and Granich right. First, access to antiretroviral therapy (ART), a treatment for HIV/AIDS, has greatly improved in parts of Africa. Second, while epidemiological models are nothing new, they are increasingly guiding strategies to curb HIV/AIDS and helping to steer future research and policy.
By looking at disease prevalence and ART availability, the new modeling analysis gives a broad picture of how close we might be to tackling the pandemic. And the models point to optimism.
AIDS remains a troubling pandemic. Last year alone, UNAIDS, the United Nations-sponsored organization which tracks global HIV prevalence and treatment coverage, tallied roughly one million deaths. UNAIDS also says that nearly 37 million people live with HIV worldwide, mostly in developing countries where accurate information about HIV, testing facilities, and treatment may not be readily available. And in some regions — including Eastern Europe, Central Asia, and parts of the U.S. — HIV is on the rise.
Despite these numbers, the global rate of new infections each year has been falling — down 16 percent among adults and 35 percent among children since 2010. In particular, HIV infection rates are decreasing in much of Africa, Western Europe, and the Asia Pacific region. More people also have access to ART than ever before.
Models can use all these data to help show possible outcomes for the disease, says Kate Mitchell, an epidemiologist at Imperial College London. To build a specific HIV model, for example, researchers could look at regional data on how often people have unprotected sex, the uptake of testing and treatment, and the current number of known infections.
“What we’re often trying to do is predict into the future,” Mitchell says, by asking: “What would happen if we keep doing pretty much what we’re doing now? What would happen if we increase testing or treatment? What would happen if people’s behavior changed in a particular way?”
The new modeling by Williams and Granich zooms in on a few key African regions, pulling data on the current prevalence of HIV as well as local policies. Specifically, they look at the availability of ART and whether the drug can help end AIDS in the regions (fully eradicating HIV is another matter, as the virus can persist for many years in someone who has it). While ART isn’t a cure, it can stave off AIDS. The drugs can also make HIV undetectable in an infected person and prevent them from transmitting the virus to others.
In nations with solid access to ART, such as Kenya, Tanzania, and Uganda, Williams and Granich think HIV infections can be brought to that key threshold — one case in every 1,000 people — by 2020. Southern African countries that have had lower ART access might expect to reach the same point by 2030. This means fewer people would die from AIDS in Africa and HIV would not be transmitted so easily, so long as people with infections continue taking the drugs. And if southern African countries increase access to ART, there will be tangible benefits: Many thousands, if not millions, of lives might be saved.
In an earlier model, published in 2009, Williams, Granich, and others projected an outcome that never happened: meeting the one-in-1000 threshold by 2016. In this model, they looked at the same parameters — HIV prevalence and ART availability — but expanded it across the world, rather than focusing on smaller regions. They also assumed a hypothetical strategy with wider distribution of ART than actually happened. Other researchers called the work “blue skies thinking,” and while the “precise predictions were very optimistic,” says Mitchell, the model helped change “people’s opinions about what we could do going forward.”
The new analysis is “carefully grounded in data,” adds Mitchell, who was not part of the research team. What’s new about the latest effort is that it focuses on key nations in Africa where the availability of ART has improved and where the cost has come down. The researchers also compared the model projections to the actual rate of new infections and found they were largely in agreement.
But the new model still relies on an optimistic assumption: the expanding provision of ART. For some regions, this will be possible. In eastern Africa, many countries are already at the one-in-1000 mark or on track to meet it. South Africa has offered ART to all people with HIV since 2016 and now has more patients on ART than any other country. But such interventions cost money, and other southern African nations, such as Swaziland, have limited budgets and have been relatively slow in tackling HIV/AIDS.
Still, as with the 2009 paper, the new model could push policy in a positive direction. According to Williams and Granich, the global annual cost of HIV/AIDS treatment is approximately $40 billion. If public health officials can get more people on ART, keep the remaining 35 million HIV-positive individuals on the drugs, and get the cost of those drugs to $100 per person per year worldwide, that cost would drop to $3.5 billion.
That’s all very well, says Anthony Fauci, director of the National Institute of Allergy and Infectious Diseases (NIAID) in the U.S., but he points out that models can only ever be as good as the assumptions or data one puts into them. While models can inform decision-making, Fauci adds, public health agencies must also take a pragmatic approach — and pull even more data from the ground.
Despite the limitations, some models do reflect reality — and influence policy. Models have, for instance, helped improve guidelines on ART distribution — policies that will be key in realizing the projections made by Williams and Granich. In 2014, Mitchell co-authored a major study that looked at what might happen if a broader group of HIV patients in South Africa, Zambia, India, and Vietnam had access to ART as a result of relaxing guidelines for initiating treatment.
As their disease progresses, people with HIV have lower numbers of certain types of white blood cells that are essential to the human immune system and the exact count can help gauge the severity of their infection. Previously, guidelines from the World Health Organization (WHO) said only people with HIV who had a white blood cell count of 350 or lower should have access to ART. Mitchell and her colleagues modeled the effects of raising the threshold to 500 or removing it altogether. They found that the shifts were both cost-effective and provided significant health benefits. The WHO now recommends that countries offer ART to all people with HIV, regardless of their white blood cell count.
Andrew Phillips, an epidemiologist at University College London, says the paper influenced the policy. “It would certainly not be right to suggest that modeling has been the only reason for introduction of these policies,” he says. “But I think it has been one essential element.”
In some cases, data can provide unexpected insights into a new strategy. Bershteyn, of the Institute for Disease Modeling, and her colleagues, modeled the policy of offering ART to everyone who is HIV positive, regardless of their white blood cell count. The model showed that even if you discount the direct medical benefit of ART, which raises white blood cell counts, it was still better to offer the drugs to everyone because it meant more people would come to take HIV tests and engage with health clinics. That alone changed behavior so that more people got treatment, says Bershteyn, which “was the crucial thing.”
Insights like that can drive policymakers to take models seriously and may even help convince politicians to address epidemics such as HIV, says Rachel Baggaley, coordinator of HIV testing and prevention at the WHO.
Modeling can also allow recommendations to be neatly targeted to specific places. Take Russia, where the HIV epidemic is growing. The rate of new infections rises between 10 and 15 percent every year. Starting in 2010, a main route for transmission — injection drug use — got worse after the number of needle exchanges in the country fell from 80 to just 10 due to funding cuts. A study published in The Lancet in July used a model to explore the dynamics of HIV in Omsk, Russia. The authors found that by scaling up needle exchange programs and ART recruitment, among other things, the rate of new HIV infections in the city could be halved within a decade.
Not all of the insights gleaned by modelers get to influence policy. Baggaley points out that governments are not always willing to engage with the problem regardless of well-researched predictions. Even when the will is there, it takes a long time, and often millions of dollars, to put new recommendations into practice. Still, modeling analyses like the new one from Williams and Granich may show a way to end one of the most persistent pandemics in history.
Chris Baraniuk a freelance science and technology journalist based in the United Kingdom. His work has been published by the BBC, New Scientist, Scientific American, and The Atlantic.









Comments are automatically closed one year after article publication. Archived comments are below.
At least things are going in the right direction! I am reading a book by Ted Neill who spent time in Africa working with kids affected by HIV. His story is compelling and heartbreaking, but it is something that we need to keep in our minds so we can push for a way to keep these children from contracting this. His book is called Two Years of Wonder, it affected him so much seeing these children dying that it actually plummeted him into a deep depression which for him added insult to injury. I probably couldn’t handle see the devastation caused by this to those kids.
Fostemsavir, another drug for the treatment of drug-resistant HIV, prevents HIV from attaching to CD4 cells by sticking to the virus, not the cell. It prevents a change of shape of HIV’s gp120 protein that is necessary for it to lock on to the CD4 receptor. Fostemsavir, is not licensed yet.
In this background, can Fostemsavir be further developed to use as latency reversing agent to clear HIV reservoirs?
A Hiv prevention strategy, Is this a new story in the fight against HIV/AIDS? Since the world is striving to find new HIV treatment methods to end the pandemic permanently by the year 2030 or before it.
Various research are underway to develop either functional or permanent cure through bnAb, HIV vaccine, organ transplant therapy, Long acting ART etc so that the pandemic can be eradicated at this generation level only without buck passing the responsibility on to the next generation level.
Therefore, scientific and research oriented findings may be promoted so that new ideas and creativity may flourish to end the pandemic at the earlier.
Excellent overview of where we are and where we are going. We must always remember the lesson of polio which is that bringing the epidemic under effective control was succesfully achieved but finding and treating the last few cases was, in some ways, an even greater challenge. For HIV we have drugs that are effective, have minimal side effects, do not lead to drug resistance, render one uninfectious to others, reduce the risk of TB by about 60% and, at least in developing countries, cost about US$1.50 per week. We can and must end the epidemic of HIV throughout the world.