How a Croatian Lab Spawned a Buzzy Peptide Now Popular With MAHA
In the spring of 2024, Flynn McGuire started to notice glowing references online to an unapproved drug that proponents claimed could help the body heal more quickly. McGuire is a chief medical resident at University of Utah Health, where he works with patients rehabilitating from cancer and injuries, but he had never heard of the substance, called BPC-157. Colleagues he consulted hadn’t heard of it either. So he decided to conduct a formal review of the scientific literature in his field. “I wanted to see if there was anything to it, or not,” he said.
What McGuire and his colleagues found as they read through the literature was a decidedly mixed bag. Almost all the existing data on BPC-157 comes from a single group of researchers in Croatia. In rodents, the substance promotes the growth of new blood vessels and reduces inflammation, enhancing healing in tendons, ligaments, and muscle. Researchers have documented few side effects. But there is very little data on how the drug works in humans, and, until well-designed human studies are conducted, McGuire and his co-authors concluded, the substance should be approached cautiously. In an email to Undark, McGuire was blunter: BPC-157, he wrote, “Should not be used by humans.”
“The amount of hype to evidence is just so skewed, it’s crazy,” McGuire told Undark in an October interview. Indeed, BPC-157 is having a moment. Once the province of hardcore gym subcultures, the substance has been embraced by popular podcasters, wellness clinics, and patients seeking relief from health conditions that are not well-managed with traditional medical treatment.
BPC-157 is a kind of chemical called a peptide, and the U.S. Food and Drug Administration treats it and some other peptides as unapproved drugs — going so far in 2023 as to explicitly prohibit compounding pharmacies from supplying BPC-157 to patients. Health and Human Services Secretary Robert F. Kennedy Jr. has more recently signaled a desire to loosen restrictions on experimental medical treatments, including unapproved peptides like BPC-157.
These drugs have gained traction within Kennedy’s Make America Healthy Again, or MAHA, movement. At a MAHA summit held in Washington, D.C. this past November, panelist Josh Fritzler, an executive at two affiliated compounding pharmacies based in Florida, said his grandmother is taking BPC-157. The peptide works, he said, adding that “it’s just one example of these products that can change people’s lives.”
At a time of dramatic changes in U.S. health policy, BPC-157 has become a kind of test case for how President Donald Trump’s second administration will regulate experimental therapies. In Kennedy’s view, it’s not the government’s job to block access to unproven treatments. He has argued, instead, that individuals should be able to educate themselves and then make an informed decision.
In the case of BPC-157, this raises a thorny set of questions: In a world of hype and financial conflicts of interest, how informed can patients be? Once a gray market is thriving, who, if anyone, will foot the bill for rigorous research? And where did this substance even come from?
Peptides are short and simple chains of amino acids that serve as the building blocks of proteins. In the body, they often act as a communication system between cells, and scientists have long explored peptide-based therapies. Today, some peptides are widely taken as medication: Insulin, for example, is an FDA-approved peptide, as are GLP-1s, the popular weight-loss drugs.
The origins of today’s BPC-157 buzz lie in a laboratory in post-communist Croatia, and with its leader, Predrag Sikiric, a professor of pharmacology at the University of Zagreb.

Predrag Sikiric leads a lab at the University of Zagreb in Croatia that has produced almost all of the existing data on the popular peptide BPC-157.
The earliest entry for BPC-157 in PubMed, a major database of scientific papers, is a study published in 1992. Authored by Sikiric and 17 others, the study references “a new potent peptide” that the team had previously isolated from human gastric juice and dubbed Body Protection Compound for its apparent protective effects on a range of body parts. In this particular study, the researchers performed surgery on rats, removing a different organ from each rodent, in an attempt to better understand which organ systems BPC-157 interacts with outside of the stomach.
Sikiric has since published more than 150 papers on BPC-157. His team has hypothesized that the peptide plays a significant role in helping the body cope with stress, and that it may work, in part, by controlling the development of new blood vessels and decreasing inflammation.
Another group of researchers, located in Taiwan, has also conducted several studies of BPC-157. In an email, Jong-Hwei Su Pang, a professor at Chang Gung University’s Graduate Institute of Clinical Medical Sciences, wrote that the peptide might be produced by stomach microbes, as its amino acid sequence doesn’t appear in the human genome.
BPC-157 studies listed on PubMed consistently show positive results. In a review published last year, Sikiric and his colleagues describe BPC-157 as a drug like none other: In animal models, it counteracts symptoms akin to those of Parkinson’s, Alzheimer’s, and schizophrenia. It heals ulcers and skin wounds, they write — along with injured tendons, muscles, and bones. It combats dry eye syndrome and “presents prominent anti-tumor potential.”
But there are significant caveats.
For one, nearly all of the work was done in experimental animals. A crucial next step in drug development, several experts told Undark, is to test for safety and efficacy in humans. Typically, before a drug can be marketed, it must pass through a series of clinical trials, in order to identify safe dosages and to determine that any positive outcomes are due to the substance itself, and not to bias in the study design. When subjected to this level of experimental rigor, the vast majority of drugs actually fail.
“We’ve cured cancer in mice plenty of times,” said McGuire. “Haven’t done it in people yet.”

Flynn McGuire is a chief medical resident at University of Utah Health. After he noticed the online buzz around the peptide BPC-157 in 2024, he conducted a formal review of the evidence behind it. He told Undark the drug “should not be used by humans.”
Visual: Kiana Schriever
Sikiric’s recent review does reference some human studies. Most involve a relatively small number of subjects, and few of the research teams seem to have actually published papers describing their methods and data. For example, researchers have tested BPC-157 enemas as a treatment for ulcerative colitis in a pair of clinical trials conducted by the Croatian pharmaceutical company PLIVA in the 2000s. Any papers resulting from these two trials do not appear to be indexed in major global databases like PubMed, and it’s not clear if they were ever published in peer-reviewed journals.
A 2015 trial linked to Sikiric, and registered on the U.S. government website clinicaltrials.gov, was designed to test an oral version of BPC-157. According to these U.S. government records, the team submitted data to clinicaltrials.gov but then withdrew it before undergoing an outside review. Without offering a citation for a published paper or sharing any data, Sikiric’s 2025 review states that this clinical trial found that the drug was safe and well-tolerated in humans.
(Over the course of several months, Undark sent numerous messages to Sikiric. Undark also emailed several of his co-authors and officials at the University of Zagreb School of Medicine, but was ultimately unable to conduct an on-the-record interview with him or a team member. Sikiric did not respond to repeated requests to share data from the trial. Undark sent additional queries to the trial’s principal investigator and to the hospital in Tijuana where it was scheduled to take place, but received no reply.)
There’s also a question of undisclosed financial conflicts of interest.
Sikiric is named on BPC-157-related patent applications dating back to at least 1989, and Croatian government records list Predrag Sikiric as an owner of PharmaCotherapia, the company that sponsored the Tijuana trial. Additionally, Sikiric is listed online as the CEO of a company called Diagen, which says it owns a patent for a “special stable version” of BPC-157. The patent is currently for sale on the company’s website. These conflicts were not disclosed on any of the team’s published papers that Undark reviewed.
Over the past couple of years, several reviews have been published in academic journals, by researchers at major universities in the U.S., Britain, and Poland. Most concluded that the substance appears quite promising, but that there are too many unknowns to justify its clinical use at this time.
A review team from Poland, for example, suggested that future studies should focus on identifying possible side effects. It’s plausible, they wrote, that new blood vessels could accelerate cancer by carrying extra oxygen to tumors.

In a dissenting comment, Sikiric and colleagues said that BPC-157 had been “unjustly accused of potentially harmful effects.” They pointed to a list of research findings that, in their view, rule out the possibility that the peptide causes cancer and other potential harms.
The Polish team replied with new critiques of the Croatians’ research. The vast majority of the roughly 200 BPC-157 studies listed on PubMed include either Sikiric or a colleague, Sven Seiwerth, as a main author, they noted. Among other things, they wrote, this could lead to confirmation bias. And absent more rigorous studies, the team warned, “it will remain difficult to convince the broader medical community that this so-called ‘wonderful’ compound meets the criteria of a legitimate therapeutic agent.”
But it turns out, one group had begun to give it a go anyway.
Seeking to improve their performance, bodybuilders and other hardcore gym users had started to network with each other on burgeoning internet forums in the early 2000s. On sites with domain names like Bodybuilding.com and ThinkSteroids.com, athletes swapped tips and shared personal experiences. Luke Turnock, a criminologist at the University of Lincoln in the U.K. who studies enhancement drugs, told Undark that medical research was woven into those conversations.
Initially, the bodybuilders’ focus was on anabolic steroids, he said. But around 2010 — as Sikiric was conducting rat studies — peptides came onto their radar. Since most peptides were not approved, it was illegal to sell them as medicines in some countries, including the U.S. and the U.K., said Turnock. Sellers have attempted to take advantage of a perceived legal loophole, labelling the substances as “research chemicals” that are not for human consumption.
Hardcore gym-goers initially sourced their unapproved peptides from laboratories in China, said Turnock. Like insulin and other peptides, they typically had to be injected. As word spread, and demand grew, Turnock said, wholesalers in the U.K. and likely the U.S. started to import the substances and set up their own websites. Soon, the gray market expanded beyond gym subcultures. By 2018, for example, there was a lively Reddit community focused on peptides.
Sikiric and colleagues said that BPC-157 had been “unjustly accused of potentially harmful effects.”
“Everybody kind of knew they were doing something a little mad scientisty,” one Reddit user told Undark in a Zoom interview. “We were looking at these rodent studies, and we’re like, ‘these look really promising.’ And we’re doing the math on species conversions to figure out human doses.” Nobody was under the illusion that what they were doing was definitively safe, he continued. Many of the bodybuilders on the Reddit forums were already using anabolic steroids for performance enhancement, he recalled: “To them, throwing in some peptides is like baby stuff.”
The Reddit user had a different reason for trying BPC-157. He had been diagnosed, he said, with hypermobility spectrum disorder, a condition in which one’s joints move beyond normal range. It caused him severe chronic pain, which at its worst, made it impossible to even walk around the block. He wanted to fortify his body by strengthening the muscles around his joints, but he was so injury-prone that workouts often left him feeling worse. Doctors didn’t have much to offer, he said, so he decided to research other options and eventually settled on several peptides, including BPC-157, which he took for over a year. During that time, exercise became easier and the pain went away.
The Reddit user asked to remain anonymous because he now does freelance science writing — a door that opened for him, he said, after he combed through the scientific literature and wrote up his findings in a 57-page Google Doc called the “Peptide Primer,” which is online and free for anyone to peruse.
Among the highlights: a manuscript detailing the clinical trials of BPC-157 as a treatment for ulcerative colitis. A member of the research team must have passed it on to somebody else, the Reddit user surmised. “I kind of got the impression that they were doing me a solid by putting my mind at ease,” he told Undark — and he said that he appreciated the additional details showing that the drug was safe and well-tolerated, at least in these small studies.
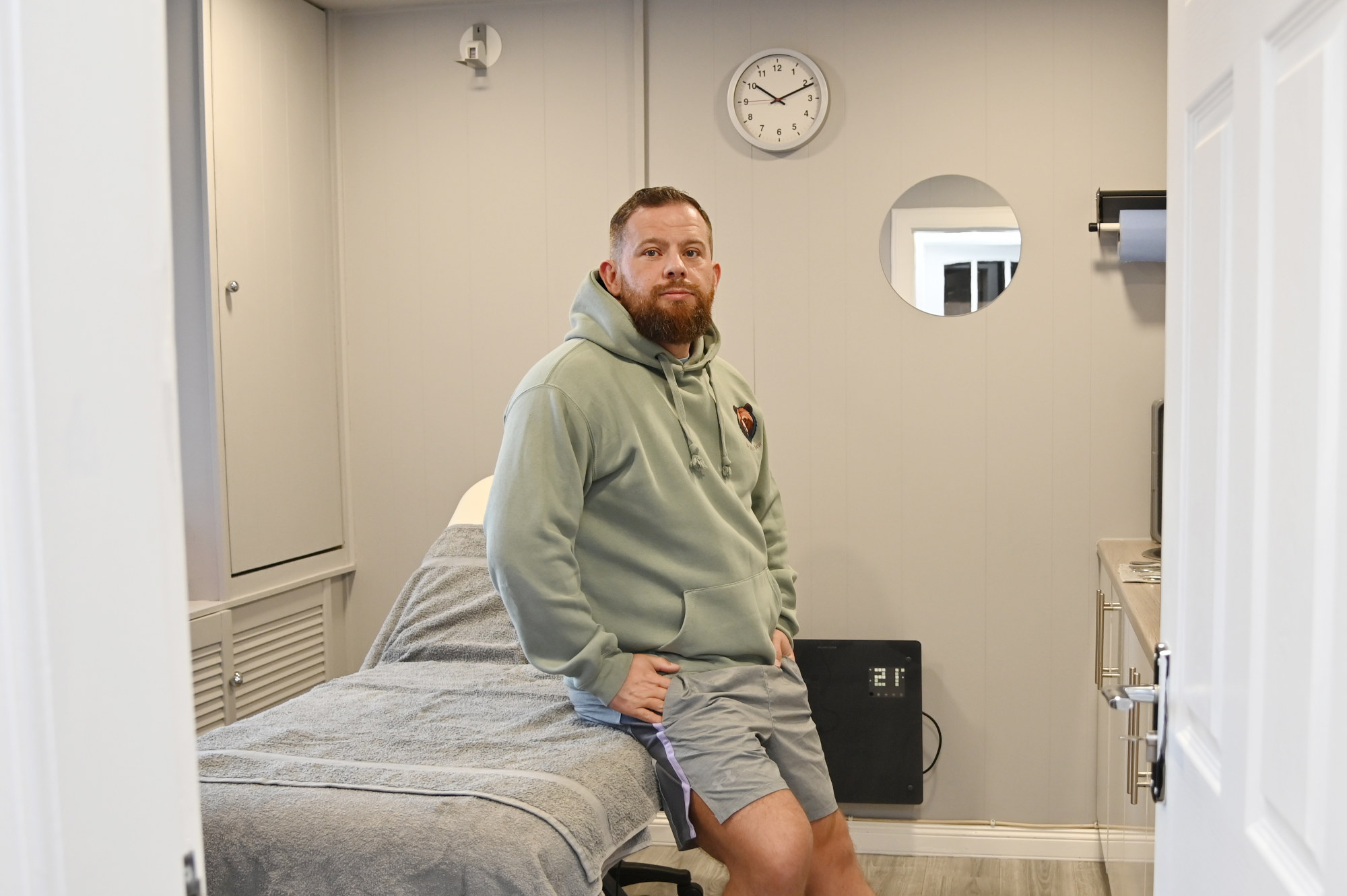
A Reddit user in a BPC-157 forum, Ryan Beattie, told Undark that he decided to try the peptide last year in an effort to live a healthier lifestyle while weaning off the powerful painkillers he had been taking for more than a decade. Beattie is a former rugby player who owns an injury rehab clinic near his home in Scotland. (Since his first interview with Undark, he has also taken a position with a company that sells peptides). Among other things, he credits BPC-157 with helping to alleviate his struggles with pain after working out in the gym, and he’s been able to significantly reduce his use of prescription painkillers. He acknowledges, though, that the dearth of human research makes it difficult to rule out the placebo effect.
Not everyone on Reddit has had a great experience with BPC-157. Some users report side effects: an itch that spread over one person’s entire body; intense anxiety (“It felt like someone was grabbing my chest and squeezing it,” one user wrote. “I could not even close my eyes because I would then begin hallucinating …”); and the inability to experience joy or pleasure, known as anhedonia. As with the purported benefits of the peptide, it can be hard to determine what link — if any — exists between BPC-157 and these bad experiences.
Dosing can be tricky, too. Because BPC-157 is a research drug, Beattie decided to aim for the most impact at the lowest dose. But he’s seen people post about taking what he characterized as “silly, silly amounts.”
Several of the experts who spoke with Undark expressed additional concerns about the purity of substances purchased from underground websites. “You don’t even know what you’re buying inside that bottle,” said Matthew Fedoruk, chief science officer for the U.S. Anti-Doping Agency. “It could be a peptide. It could be a steroid. It could be something just like water.” Consumers risk injecting themselves with something that is actually quite dangerous, he cautioned.
Some of these issues could be alleviated with robust human-based research, followed by FDA approval if warranted. Here, however, the gray market may be serving as a significant obstacle.
For one, it makes a robust clinical trial logistically tricky, said Christopher Milne, past director of research at the Tufts Center for the Study of Drug Development. Before approving a new drug, the FDA typically requires companies to conduct a type of study called a randomized controlled trial, in which study participants are allocated by chance to groups that either receive the drug, or are administered a placebo. People might prefer to get BPC-157 on the gray market, rather than risk being assigned to the placebo group. And even after people enroll, Milne wrote in an email to Undark, the substance’s short half-life would make it difficult for the researchers to ensure that participants are sticking with the trial protocol and not supplementing with gray market BPC-157.
“Everybody kind of knew they were doing something a little mad scientisty.”
Even absent these logistical hurdles, pharmaceutical companies might not want to invest in clinical trials. Developing and testing a new drug is expensive, and companies count on recouping their costs by having, in effect, a monopoly on the market for a new drug. Paul Knoepfler, a stem cell researcher at University of California, Davis School of Medicine, has noted that this financial incentive may be diluted by “the fact that potentially hundreds of thousands of people are already taking these peptides.” So, the company would have to compete against unauthorized sources that haven’t invested anything into research.
For a glimpse of what happens when a peptide becomes popular before it’s been fully developed by a pharma company, consider retatrutide. Over the past year, this GLP-1 has become a hit among gym-goers, even though it’s still being vetted in clinical trials overseen by Eli Lilly.
The FDA has been sending warning letters to the companies that are illegally supplying the peptide, said Tenille Davis, a pharmacist and chief advocacy officer for the trade group Alliance for Pharmacy Compounding. The agency hasn’t been able to make a dent, she observed, as new websites continue to pop up. “Honestly,” she said, “I feel bad for the FDA, because they’re playing a game of Whac-a-Mole.”
Although it’s legally risky to prepare and sell BPC-157, some pharmacies and clinics do offer it to patients. In fact, the FDA’s regulatory actions have sparked vocal resistance from people who support the clinical use of peptides, and who say that current restrictions infringe on the rights of patients.
One leading peptide advocate is Edwin Lee, a Florida endocrinologist. Lee told Undark that he learned about peptides in 2016, while attending a conference. He decided to introduce two of them, BPC-157 and thymosin alpha 1, into his clinical practice. (While not approved in the U.S., thymosin alpha 1 has been approved in numerous other countries, principally as a treatment for hepatitis B and C.) Lee’s patients responded well to peptides, he said, “so I experimented with others.”
He also began to build a national reputation: Lee is a co-founder of the Clinical Peptide Society, which offers certification classes, and in 2019 he published a book titled “The Fountain of Youth With Peptides.”
At his fee-for-service clinic in Orlando, Lee writes peptide prescriptions for patients who want to use the substances with physician oversight. That prescription goes to a compounding pharmacy, which then ships the peptides to the patient. Compounding pharmacies are regulated at the state and federal levels and subject to government inspections, making them a safer option than unlicensed online vendors, said Lee.

For the compounding pharmacy, this is not without legal risk. In late 2023, the FDA added more than a dozen peptides — including both BPC-157 and thymosin alpha 1 — to its list of substances that should not be compounded because the agency has identified “potential significant safety risks.” For BPC-157, the agency warned that the peptide could trigger an immune response. The FDA also said that drugs containing BPC-157 might harbor impurities, and that the peptide lacks complete safety-related information.
Today, it is “likely not legal” for compounding pharmacies to provide BPC-157, said health care attorney David Holt. (Expert opinion varies over what degree of legal risk — if any — physicians face for prescribing peptides like BPC-157. In an email to Undark, Lee wrote that the FDA “has not prohibited physicians from prescribing these agents.”)
So far, though, the FDA appears to have taken few steps to discipline those pharmacies that have continued to sell BPC-157, according to a government database that contains warning letters sent to companies it believes are violating federal guidelines. “Some of these compound pharmacies, they know the rules,” said Lee. “But they’re basically saying, ‘Look, we believe that peptides will be saved.’”
In fact, pharmacists have pushed back against a regulatory process they describe as murky and unfair. The peptides “should be re-evaluated under a better process by the Pharmacy Compounding Advisory Committee,” said Davis, referring to a panel that advises the FDA on issues related to drug compounding. Ideally, wrote Davis in a follow-up email, the government agency will spell out its specific concerns about each peptide, and then describe what data would be needed for the FDA to overturn its prohibitions.
“Some of these compound pharmacies, they know the rules. But they’re basically saying, ‘Look, we believe that peptides will be saved.’”
For now, at least two lawmakers — Tommy Tuberville and Diana Harshbarger — have issued letters asking the FDA to limit enforcement action against certain peptides, including BPC-157. Under such a policy, the FDA would not penalize pharmacies for compounding the substances.
(An HHS spokesperson said the FDA would not comment on the agency’s enforcement policies for BPC-157 and other unapproved peptides.)
For his part, Lee founded the nonprofit Save Peptides, which has launched a petition asking lawmakers to intervene. It currently has more than 9,000 signatures. Many of the signatories have left messages crediting peptides with improving their health. Some messages carry a decidedly anti-government tone: “THE FDA IS A ROGUE CORRUPT ORGANIZATION AND MUST BE CHALLENGED ON EVERY DECISION,” reads one message from nine months ago.
While the petition’s grievances are specific to peptides, they reflect long-running criticisms of the FDA, which has often come under fire from patient advocacy groups who want better access to experimental treatments that might ease the progression of deadly diseases.
In an effort to encourage the FDA to change its policy, Lee has conducted a series of studies testing BPC-157 on the patients in his clinic and at one other clinic in Florida. So far, he has published three papers in Alternative Therapies in Health and Medicine, a peer-reviewed journal devoted to complementary and alternative medicine, such as acupuncture and chiropractic. Lee’s studies are smaller than most rigorous clinical trials, ranging in size from two to 16 study participants, and don’t include comparison groups. (Lee told Undark that more research is underway, including one larger study and another with a comparison group.) “If we do some certain studies,” he said, the FDA may say, “‘you’ve shown safety and efficacy.’” The agency, he added, might then allow the peptides to be compounded: “That’s all we’re asking.”
Many are hopeful that Robert F. Kennedy, Jr., who oversees the FDA in his role as secretary of the Department of Health and Human Services, will change the policy.
In a May interview with wellness entrepreneur Gary Brecka, Kennedy elaborated on how he sees the role of the FDA with regards to experimental treatments, including peptides. “Our position is that FDA has a job, which is to do the science on these kinds of issues and then tell the public what they’ve learned from the science,” he said. It’s not the FDA’s job, he insisted, to dictate what physicians can prescribe or what patients can take. If a person wants to take an experimental drug, he continued, that person should be allowed to do that.

In December, wellness entrepreneur Gary Brecka, who promotes BPC-157 on his website, hosted FDA Commissioner Marty Makary in Miami. On Instagram, Brecka posted this image showing Makary trying out a cold plunge, which Brecka also markets.
Visual: Screenshot taken by journalist Sarah Karlin-Smith
“You’re going to get a lot of charlatans, and you’re going to get people who have bad results,” Kennedy acknowledged to Brecka. But the status quo, he argued, is no better. “Leaving the whole thing in the hands of pharma is not working for us.”
Many MAHA-aligned people pushing for reforms have a financial stake in the outcome.
On his website, Brecka promotes a BPC-157 nasal spray blend for $375. A separate BPC-157 blend, sold as patches, costs $575 for a one-month supply. In December, Brecka hosted FDA regulators in Miami. The agency’s commissioner, Marty Makary, and deputy commissioner for Human Foods, Kyle Diamantas, arrived wearing suits, said Brecka during a MAHA Media Hub talk the following day. “I threw them both board shorts and stuck them down in a 50-degree cold plunge,” he continued. “I think I’ve turned them into converts.” (Brecka markets cold plunges, currently on sale for $6,999.)
In a Dec. 23 podcast episode, Brecka pressed Makary on how FDA policies might evolve with regard to a wide range of unapproved treatments and devices, including a blood filter, stem cells for type 1 diabetes, and peptides like BPC-157.
The commissioner’s responses were open yet non-committal. “If a therapy has good data, we’d like to see it,” said Makary. Speaking about peptides specifically, he said the agency will need to review them to determine which are “harmless” and which require additional study.
Victor Prisk, an orthopedic surgeon in Pittsburgh, said he understands the appeal of a libertarian approach to medicine, in which patients educate themselves and then make an informed choice about whether to proceed with an unapproved drug. The problem, he said, is the hype around BPC-157 can be quite misleading, particularly when it comes from his fellow physicians. Podcasters with massive followings are a problem, too, he said, naming Joe Rogan and Andrew Huberman.
In a podcast episode released in April 2024, Huberman struck a cautionary note, with the Stanford professor using the words “cancer,” “tumor,” and “risk,” more than three dozen times. Later in the year, though, he brought on a guest physician, Craig Koniver, founder and medical director of Koniver Wellness in South Carolina. The clinic website includes a page devoted to peptides, and Koniver told Huberman he would like to use BPC-157 with almost every patient. He described a BPC-mixed injection as “awesome, super-safe. And it’s amazing for people.”
Brigham Buhler, owner of a telehealth company and a mail-order compounding pharmacy, took a similar tone in a 2023 episode of Joe Rogan’s podcast. Speaking about BPC-157, he said, “it’s insane how well it works.”

McGuire, the University of Utah chief resident, suspects some of the hype comes down to pop-science podcasting and its business model. To earn money, podcasters need to release content on a regular cadence. Someone may start out discussing interventions with solid evidence behind them, “but if you got to come out with a podcast a week, at some point, you got to start reaching further and further afield for new topics or new exciting things,” McGuire observed. “Science and medicine just don’t move that fast.”
Some clinics have also promised dramatic results from BPC-157. Undark asked Holt and Nathan Downing, an attorney with Gardner Law who specializes in FDA law, to review several examples of language used by clinics. Both flagged some examples as posing potential legal risks, under statutes that limit the claims health care providers can make about drugs that are not FDA-approved. The website of one clinic, based in Utah, “makes bold and wide-ranging claims of efficacy,” wrote Downing, noting that “the government could object.” Looking at the same website, Holt also saw issues: “Most of their claims are flat-out illegal.”
For their parts, both the anonymous Reddit user and Beattie said that while they were glad to have the freedom to decide for themselves whether to take BPC-157, the hype around the product was unhelpful.
“There needs to be more research,” said Beattie. Among other things, he noted, this could help establish a recommended dose. He wondered aloud how U.S. compounding pharmacies are approaching the question of dosing, given the lack of human studies and the fact that starting on GLP-1s entails careful titration.
“You’re going to get a lot of charlatans, and you’re going to get people who have bad results,” Kennedy acknowledged.
And people with debilitating conditions aren’t necessarily expecting a miracle drug, said the anonymous Reddit user. Even FDA-approved medicines have side effects, sometimes serious ones, he pointed out. “Accepting some unknowns in terms of safety is reasonable for life-changing medication. We do it all the time.”
Meanwhile, at the MAHA summit in Washington, D.C. in November, the mood was buoyant.
“I think the future is bright with peptides,” said Buhler, who moderated a discussion on compounding pharmacies. “The administration is aware of the issue and has thankfully had tons of meetings and — ”
“Who wants peptides?!” interjected a panelist.
The audience clapped and whooped.











This article explained BPC-157 very clearly. It’s surprising to hear that it is being widely sold on the U.S. gray market despite the lack of clinical trial data and without FDA approval. It might be worth suggesting to Dr. Sikiric that he consider filing patent infringement lawsuits against U.S. sellers. If that were to happen, it could potentially serve as a catalyst for a company or research institution in the United States to initiate clinical trials.