In Rwanda, ‘Human Bait’ Volunteers Help Battle Malaria
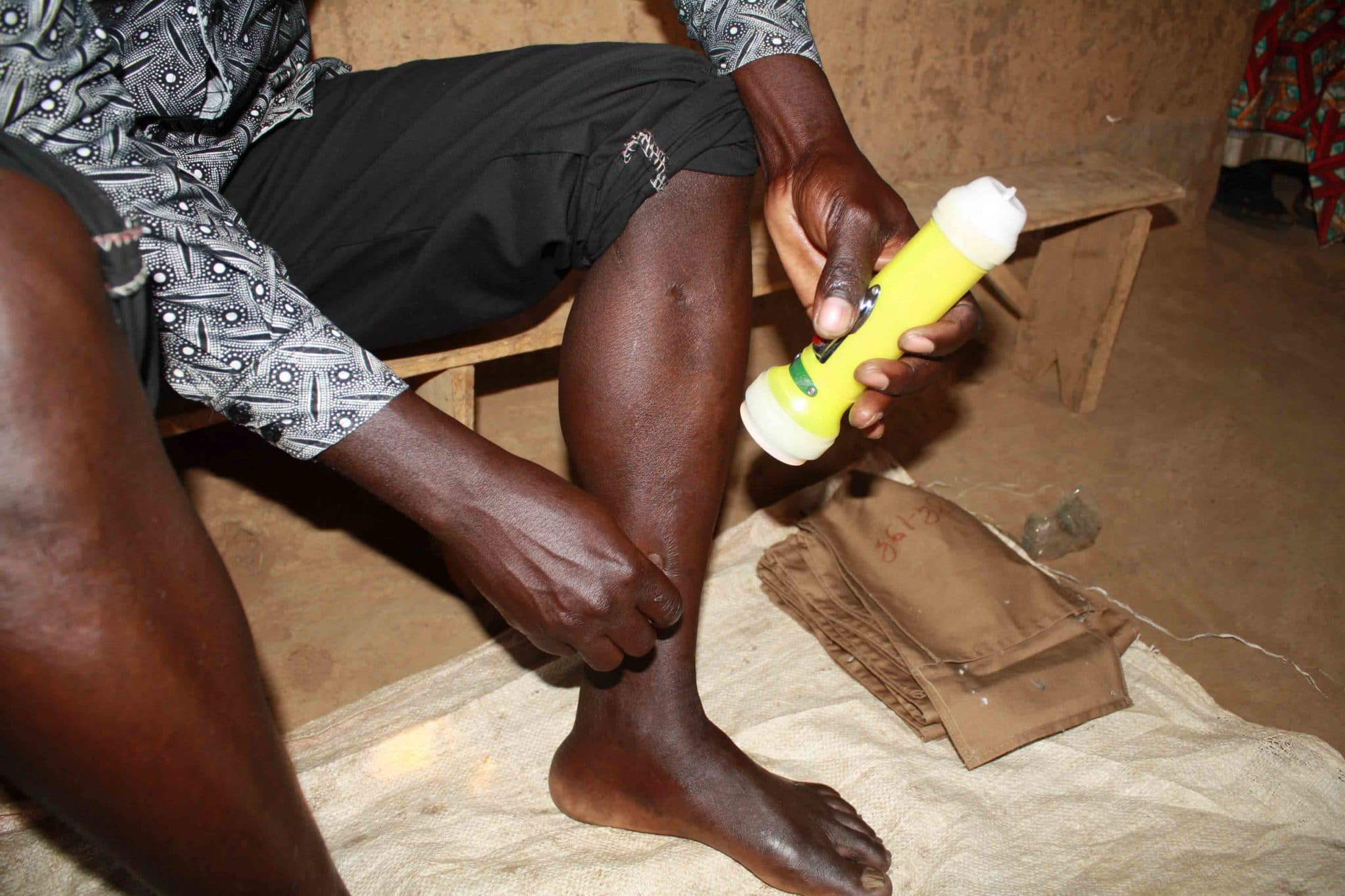
As the late afternoon sun hovers over the horizon in the Rwandan village of Mareba, about an hour from the capital of Kigali, Innocent Bizimana prepares for the mosquitos to arrive. He settles onto a bench inside his neighbor’s house, spreads a tarp beneath his bare feet, and rolls his pant legs above his knees. He arranges little glass vials, empty and clinking, on the tarp around him, along with tufts of cotton that will serve as stoppers.
And then he waits for darkness.
Bizimana is one of 12 villagers who are paid about $3 a night to serve as bait for mosquitoes, which they trap and transfer to technicians at a nearby entomology lab that serves the whole district of Bugesera, one of the most malaria-prone regions in Rwanda. Many of the mosquitoes carry Plasmodium falciparum, a species of malaria parasite capable of killing within 24 hours of the first sign of symptoms if not treated.
Bugesera’s human bait volunteers are on the front line of malaria research in Rwanda, one of only four high-burden countries — along with India, Ethiopia, and Pakistan — that managed to see a significant decline in malaria in 2017. All four relied on similar strategies, according to Ren Minghui of the World Health Organization, writing in the journal Infectious Diseases of Poverty last year: preventing the spread of malaria with bed nets and pesticides; rapidly testing and treating malaria cases; and “significantly” strengthening malaria surveillance.
Rwanda, along with a handful of other countries, has established a strong entomological surveillance program. Health officials don’t just monitor the places where people become sickened with malaria; they keep track of the mosquitoes themselves. “They have made huge progress in going toward malaria elimination,” says Arjen Dondorp, the head of malaria research at the Mahidol-Oxford Tropical Medicine Research Unit in Bangkok, Thailand, and a member of the commission on malaria eradication convened by the British medical journal The Lancet and the University of California, San Francisco in 2017.
Over the past 15 years, since the start of an ambitious national plan funded in part by the United States and international partners, Rwanda has reduced malaria deaths by 80 percent. Child and maternal health in particular have improved significantly because of a strong focus on preventing, diagnosing, and treating cases among pregnant women and children under five. This has been accomplished in part by strengthening the country’s entire health system, and also by treating malaria as quickly as possible, distributing insecticide-treated bed nets, and spraying insecticides on the walls of homes in the regions at highest risk of the illness.
In addition, entomological “sentinel sites” like the one in Bugesera — one of 12 throughout the country — help researchers and health officials monitor malarial mosquitoes in real time. They study everything from the effectiveness of certain insecticides to the subtle genetic changes mosquitoes undergo, enabling the government to better develop and adapt its policies. And while there are risks, recruiting people to serve as bait is seen by researchers as necessary to fully understand the behavior of mosquitoes; volunteers are fully informed about potential dangers and have access to timely treatment should they be infected with malaria or some other disease in the course of their work.
Despite the progress, however, there are still around 4 million cases every year in a country of more than 12 million people. “Everyone knows how to prevent malaria, but we still have cases,” says Dr. Diane Gashumba, Rwanda’s former minister of health. (She was recently forced to resign by President Paul Kagame.) “Our wish is to have zero deaths due to malaria.”
While the Covid-19 pandemic has triggered lockdowns in Rwanda and other malaria-prone countries in Africa, last month the World Health Organization stressed “the critical importance of sustaining efforts to prevent, detect, and treat malaria” in the region during this time.
Bizimana and his fellow volunteers work six-hour shifts two nights per month, from 6:00 p.m. to midnight or from midnight to 6:00 a.m. Each hour, to avoid getting sleepy, he swaps places with the person sitting outside under leafy banana trees waiting to conduct the same work. The volunteers are not supposed to use cellphones or have visitors; Bizimana sits in his neighbor’s house so that his children — he has four — won’t interrupt. The busiest time is in the middle of the night, usually between midnight and 3:00 a.m.

But at dusk the mosquitoes are already beginning to buzz through the window and land on Bizimana’s exposed skin.
Even when it’s pitch dark, Bizimana can feel when a mosquito has landed. Without moving his leg, he switches on his flashlight, aims it at the insect, and traps it with an empty vial placed on his skin. Panicked, the mosquito flies into the vial, and Bizimana covers the top with his thumb and then plugs it with cotton.
He will do this again and again as the night goes on, placing each vial in a sack marked with the hour. In the morning, the village coordinator will deliver all the sacks, filled with dozens of vials clinking like coins, to the local lab, where an entomology technician uncaps them and places the specimens under a microscope. In the past four years, the technicians have noticed something startling: The main type of mosquito found around Bugesera is changing — and so is its behavior.
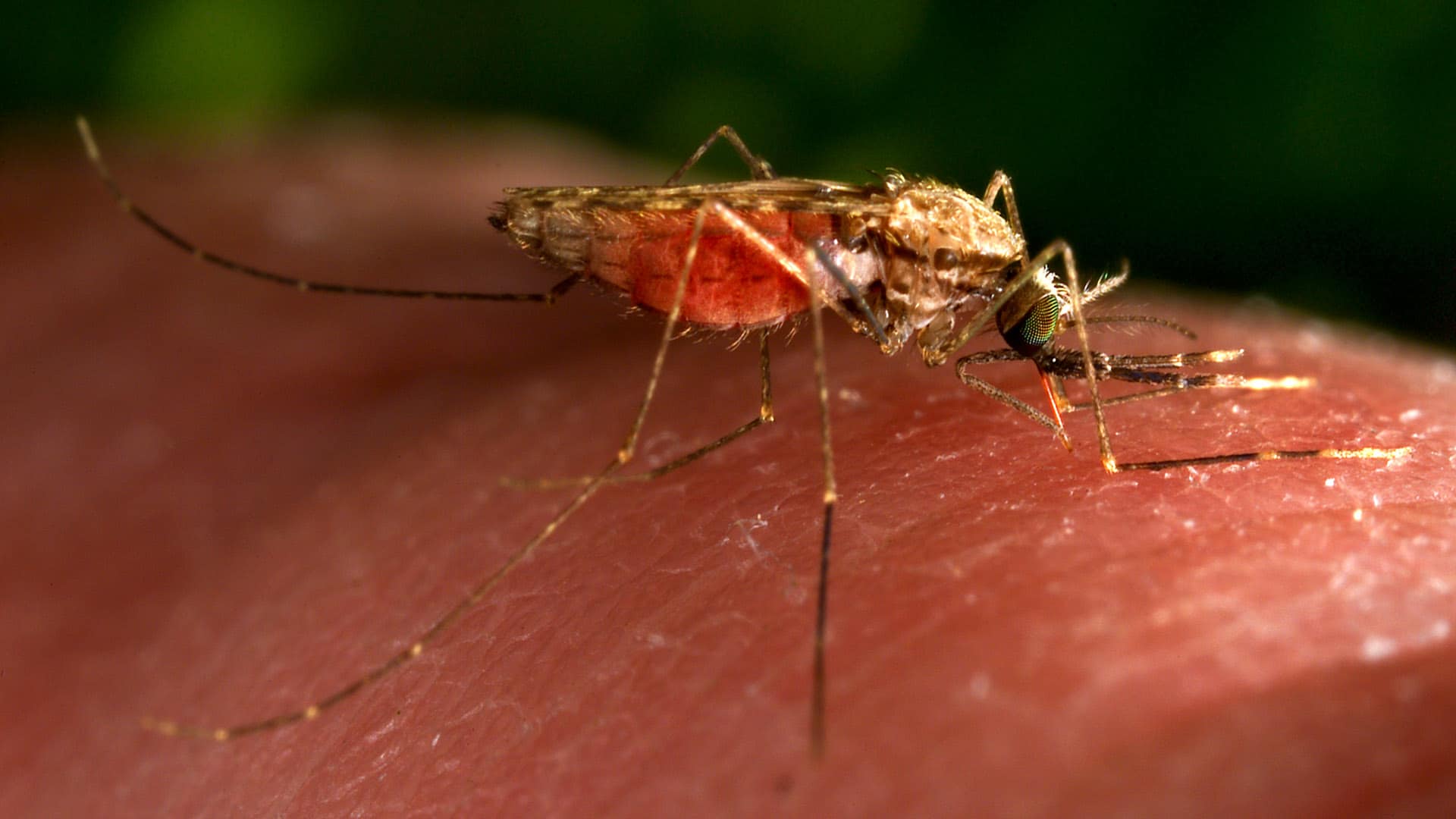
Mosquitoes are the deadliest creatures on earth. There are some 3,500 species worldwide, more than 800 of which are found in sub-Saharan Africa. They often carry fatal diseases — including malaria, which killed 405,000 people and sickened millions more in 2018. The U.S. Centers for Disease Control and Prevention estimates that nearly half of the world’s population is at risk for the disease, but nearly all deaths occur in sub-Saharan Africa, home to 10 of the 11 countries (the other being India) with the highest prevalence of malaria.
Since the turn of the millennium, the world has made substantial progress in battling the disease. According to the World Health Organization’s most recent report on the disease, in 2018 global malaria cases fell by nearly 30 percent and deaths were cut by 60 percent compared to a 2000 baseline. In the past few years, though, experts say progress has stalled. And they don’t know why.
“We need to investigate more,” says Dondorp. “Whether it’s the health system, or the insecticide resistance,” or the movement of people “because of humanitarian crises in different places in Africa.”
Last fall, the Lancet commission issued an ambitious malaria eradication plan, arguing that the disease “can and should be eradicated” within the next 30 years, especially given emerging tools to fight the disease, including entomological research and new surveillance techniques.
In many places, simply using existing tools — bed nets, medicines, pesticides — is enough to eradicate malaria, says Dondorp. But there are some places where mosquitoes are especially widespread, virulent, and efficient in the way they feed. Part of the battle is understanding how mosquitoes’ behavior — who and what they bite, and when and where they do it — is changing.

Until now, most strategies for combating malaria have depended on responding to certain mosquito behaviors: When they bite indoors at night, people use bed nets while they sleep; when the insects rest on walls to digest blood meals, the walls are sprayed with insecticides.
But a growing body of research indicates that mosquitoes are adapting their behavior — feeding at different times of the day or switching to outdoor feeding — apparently in direct response to insecticides. At the same time, in places like Rwanda, new species that are more resistant to current insecticides are moving in to replace dwindling mosquito populations that have been successfully controlled.
While insecticide resistance is the focus of most current public health research, behavioral changes are “potentially more challenging” to control, according to a recent report in Malaria Journal. “This ‘behavioral resistance’ will become an increasing problem,” Dondorp says. “So, then, new entomological tools become quite important.”
The Rwanda Biomedical Center (RBC) is the government agency responsible for health care delivery, education, and research. In Kigali, it operates an entire office devoted to understanding malarial mosquitoes and analyzing specimens from the country’s collection sites.
As Emmanuel Hakizimana, director of vector control and entomology surveillance at RBC, stands in a hallway between genetic sequencing labs, he gestures at rows of storage lockers full of preserved mosquitoes meticulously categorized by location and month of collection.
“We used to collect Anopheles gambiae sensu stricto,” he says, explaining that only a few years ago, this species made up about 93 percent of the mosquitoes collected. “Now, the dominant species is Anopheles arabiensis.”
The reason, he says, is insecticides.
Until recently, insecticides were highly effective. Nyagatare District in northern Rwanda, for instance, used to have the highest rate of malaria in the country, with more than 100,000 cases a year. After spraying began in 2009, that number dropped to about 10,000 a year, one of the lowest rates in the country. But while A.gambiae s.s. has been largely eradicated, A. arabiensis, which is much less susceptible to the insecticides Rwanda has relied on, has moved in to fill the gap. It’s less of an evolution, researchers believe, than an ecological shift: As one sub-species declines, another takes its place.
It’s hard to tell the difference between A. gambiae s.s.and A. arabiensis, especially among adult insects. They both feed at night, and their physical appearance is similar; it often takes microscopic analysis or even DNA sequencing to tell them apart. But when it comes to behavior, the differences are quite pronounced.
A. gambiae s.s. is a homebody. She — it’s always a she; only female Anopheles take blood meals — prefers to feed on people sleeping indoors. After taking a nighttime meal, she usually rests on a nearby wall to digest the blood that provides the nourishment she needs to produce eggs.
But A. arabiensis isn’t such a picky eater. Unlike A. gambiae s.s., she feeds on people and animals alike. In fact, 60 percent of the A. arabiensis mosquitoes captured by volunteers have been feeding on livestock, not people. That means the mosquitoes carrying malaria have more food sources and can bide their time before biting humans.
A. arabiensis also prefers to stay outdoors. She regularly bites people outside and doesn’t need to linger inside, resting on nets or walls to digest her food, and so she escapes many of the chemical traps that currently control mosquitoes.
Entomological research is “increasingly critical,” says Allison Tatarsky, co-director of the Malaria Elimination Initiative at the University of California, San Francisco. Monitoring mosquitoes closely helps researchers know whether an intervention — say, a new type of insecticide — is working, and it helps them keep ahead of rapid changes in the insects themselves.
“Arabiensis is the problem in southern Africa right now,” Tatarsky says. “If you don’t have entomologists, if you don’t have entomological surveillance, you’re not going to figure that out.” Rwanda, she says, has been a leader in this regard.
In many cases, these adaptive changes are happening too quickly for responses to keep pace. In Rwanda, for example, it only took about eight years for A. arabiensis to begin multiplying after insecticide spraying began.
The heart of the RBC’s malaria-control facility is the insectary, a separate building comprised of three small rooms. Here, larval A. gambiae s.s. mosquitoes that have not yet been exposed to the malaria parasite wriggle around in Tupperware containers. Once they hatch, they’re transferred to netted cages in another room where the researchers can study their habits.
The building is uncomfortably hot and humid, even for a near-equatorial country like Rwanda; the mosquitoes thrive best between 82 and 90 degrees Fahrenheit, and in 70 to 80 percent humidity. The windows are painted black, and the researchers control the lights carefully — 12 hours of light, 12 hours of dark — in order to keep the mosquitoes on an appropriate growing and breeding schedule.

The insects are bred here to test insecticides. “We only select females,” says Dunia Munyakanage, the vector control supervisor at RBC, as he demonstrates how they remove mosquitoes from their cages and place them against the wall. It’s easy to tell the difference between females and males, he adds: “Males, they have beards.”
Munyakanage pauses as his colleagues laugh, and then he chuckles, too. But it’s not a joke; the male’s proboscis — the snout it uses to suck nectar from flowers — is indeed feathered and hairy.
To move the females from the cages, Munyakanage uses a long, flexible tube — kind of like a snorkel. One end goes into the cage, and the other in his mouth; he then breathes in and out for a few moments. The mosquitoes are attracted to the carbon dioxide he expels and soon crowd into the tube, blocked from his mouth by a little metal screen.
Next, he fastens a cone to the wall, held in place with Scotch tape, and releases the insects into it. “We place these cones at different levels: at the bottom, in the middle, and on the top,” he says. “To see the quality of spraying.”
The mosquitoes stay in the cone for 30 minutes. If they all die, the researchers know the insecticide is working.
“We select, randomly, houses to be evaluated after spraying,” Munyakanage explains. “And then we will inspect at the interval of one month.” When the insects begin to die at a lower rate — anything less than 80 percent — they know it’s time to spray again. Typically, they expect to spray once a year, but sometimes it needs to happen more often.
The process is similar with testing insecticide-treated bed nets; researchers tape the cone to the net, add some mosquitoes and wait. This exercise helps them make sure the insecticides are still working to kill mosquitoes before resistance can set in.

At the moment, the Kigali insectary only works with A. gambiae s.s. raised from larvae. The researchers would like to expand their research to A. arabiensis, but the species is notoriously difficult to keep in insectaries, Munyakanage explains. When they tried in the past, the A. arabiensis refused to eat or reproduce, so the colony quickly died out.
Researchers would also like to keep wild-caught mosquitoes like the ones captured using humans as bait, rather than only raising non-malarial insects from larvae. Last year, they tried to keep some wild mosquitoes with known resistance to insecticides. But they, too, died off; the mosquitoes would take blood meals, but they never laid eggs. Munyakanage hypothesizes it was because their environment changed too much from the marshland to the lab.
But the researchers are determined to try keeping both types again this year, tweaking the insectary’s environment to better suit the mosquitoes. Munyakanage says this work is necessary to monitor the behavior of different types of mosquitoes and help the researchers — and government officials — respond quickly. “We cannot wait until we have seen the widespread resistance,” he says. “Then you are too late, and the damage will be huge.”
In addition, he adds, “I think it’s very important, very crucial now, to start looking for an effective vector control that will be applied to animals and also to the breeding sites — marshlands, rice paddies, cattle.”
There is already some research being conducted on medications for cattle that would kill mosquitoes — especially the animal-feeding A. arabiensis. “I think we need to apply all the weapons available,” Munyakanage says.
Another way to control mosquitoes outdoors is what’s called an attractive toxic sugar bait. Basically, it’s a trap that uses sugar to draw mosquitoes, but it also contains chemicals to kill the insects. “That’s what we need, is more and more tools for outside transmission environments,” says Tatarsky.

In the 1970s, a different species, Anopheles funestus, was dominant in Rwanda. After decades of work to control the species with insecticides, Rwanda radically reduced its A. funestus population. But when that happened, A. gambiae moved in.
Munyakanage is worried that cycle will be repeated with A. arabiensis. But he hopes that because today’s research is more advanced and systemically conducted by a government agency, there will be a shorter delay in taking action.
“The vector control, all the research we do, all the surveillance we do, are done under the malaria program,” Munyakanage says. “It’s not done by any private or any separate organization” where “maybe the data will come like five years later.”
“That is helping us even to mobilize funds, even to talk to the donors, even to go to the government and say, ‘Look, if you have this, we can be able to control malaria in Rwanda,”’ he says. (Last October, donors to the Global Fund to Fight AIDS, Tuberculosis, and Malaria pledged $14 billion over the next three years, but it remains to be seen how the Covid-19 pandemic will affect research operations and funding going forward.)
In Mareba, Innocent Bizimana says he’s proud to help monitor malarial mosquitoes in his village. “We are helping the government to do research on the mosquitoes,” he says, sitting up straight on the hard wooden bench in his neighbor’s house.
He also says he has become a respected figure in the village, which allows him to share what he’s learned with his neighbors and family. He makes sure there’s no standing water in his yard where mosquitoes might breed, and his household uses bed nets.
“All the training I have helps me to protect my family,” he says.
Melody Schreiber is a freelance journalist who reported from Rwanda on a fellowship with the International Women’s Media Foundation (IWMF).











Comments are automatically closed one year after article publication. Archived comments are below.
Has anyone in Rwanda looked into the AGO traps the CDC developed to help control mosquitoes in Puerto Rico?
They are patented, but I’m sure CDC would license to Rwanda for non or nominal fee. They are also really simple to make and use.
https://www.ncbi.nlm.nih.gov/pubmed/24605464