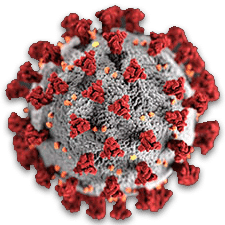
Where did the Covid-19 pandemic come from? Almost since the beginning of the outbreak, a bitter and explosive controversy has raged over the origins of the novel coronavirus known as SARS-CoV-2. The rapid shut-down of the Huanan Seafood Wholesale Market in Wuhan immediately suggested to Western observers that the Chinese government itself thought that the market was the source, especially since 26 out of 47 of the original cases could be linked to it. An article published in Nature in March 2020 seemed to leave no doubt: The virus’s genome showed every evidence of natural origins.
But the story did not stop there. Many writers and researchers suggested that the presence of a high-containment laboratory in Wuhan, the Wuhan Institute of Virology, could point to a laboratory origin for the pandemic: a bioweapons experiment; or gain-of-function research, in which genetic manipulation adds some new feature to an existing germ; or simply the laboratory escape of a lethal bat virus. Since many lab escapes have happened in the past, some argue, a lab leak is a plausible explanation for this devastating explosion of disease. While a team convened by the World Health Organization declared in March that a lab leak was “extremely unlikely” and suggested wildlife farms that supplied the market could be the culprit, a new group of scientists is now set to revisit the issue.
Still, there remains, as of this writing, no physical evidence linking the pandemic’s origins with a laboratory escape. And furthermore, from a logical and evolutionary viewpoint, there is something fundamentally wrong with all lab-leak arguments. SARS-CoV-2 is a human-adapted virus capable of effective, stealthy transmission from person to person. Lab escape theories cannot clearly account for the adaptation of the virus to its new host, or, in other words, for the evolution of human-to-human transmissibility.
In order for a virus to adapt to a new species, it needs to evolve to a point where it can easily and readily spread within that species. This is not the work of an instant, but rather the end result of a long chain of adaptation and transmission. That’s an evolutionary process. Human-to-human transmissibility has never been produced deliberately in laboratory experiments because no one knows exactly how to make a virus more transmissible among people. It’s not something that can happen accidentally, because the genetics of transmission are so subtle and complex — the result of numerous specific tiny adaptations. And a virus that’s readily transmissible among humans in the way that SARS-CoV-2 is has never been found in the wild, because animal viruses are adapted to their own host species. To make a human-to-human transmissible disease, you need human beings, a lot of human beings, to be exposed to a pathogen. And you need the repeated action of natural selection on the pathogen spreading among those human beings.
Transmission is key to a pathogen’s adaptation. In SARS-CoV-2, transmission is effective, silent, and relentless because the virus replicates at high levels in the upper respiratory tract, making it easy to spread through coughing, sneezing, talking, and breathing. According to coronavirus expert Susan Weiss of the University of Pennsylvania, SARS-CoV-2 replicates better at slightly lower temperatures than some other viruses, allowing it to populate nasal passages and the upper airways, where the temperature is lower than in the lungs. Though it has not been proven, Weiss said it would make logical sense that better replication at lower temperatures could permit the virus to shed early in the infection, before symptoms set in.
In contrast, highly pathogenic H5N1 avian flu, for instance, never acquired the ability to transmit from person-to-person at all, despite a fair number of fatal human infections. This is, at least in part, because the virus attaches to receptors deep in the lungs, and not, unlike SARS-CoV-2, in the upper airways.
How would you design a virus to spread stealthily in the ways that SARS-CoV-2 does, either for general research or for nefarious purposes? You wouldn’t. You wouldn’t know how. “There’s a vanishingly low likelihood that you could design a virus so that it spreads asymptomatically,” says Weiss.
Transmission is a subtle thing, involving many genes and many functions. Only natural selection, in the context of repeated spread from host to host within a single species, can guide its evolution. The idea that all of these traits could be accidentally picked up in laboratory experiments and introduced into a bat virus seems no more likely than the idea that they were consciously designed by researchers. Huge mink farms in Denmark and the Netherlands, where, in several instances, infected workers introduced SARS-CoV-2 to the crowded animals, show us how this adaptive evolutionary process works: The human-adapted virus rapidly evolved, several times over, to be a mink-adapted disease which may be better at transmitting among minks than people.
No one really understands the genetics of transmissibility for any virus. The closest scientists have come is in a notorious series of experiments, the results of which were published in 2012. Two laboratories, one in the Netherlands, one in Wisconsin, separately showed that by changing one aspect of transmission, the receptor by which highly pathogenic H5N1 bird flu attach to cells in the respiratory tract, they could, by repeatedly passing their altered strain through ferrets, ultimately infect ferrets via the airborne route. Many scientists insisted that this gain-of-function research was inherently dangerous, and the labs agreed to a voluntary year-long moratorium.
But as Columbia University virologist Vincent Racaniello points out, that work actually did not produce a more dangerous virus. By shifting receptors to those high in ferrets’ airways, the virus lost its virulence. None of the ferrets died. Weiss quips that sometimes gain-of-function research actually involves “loss of function.”
The closest anyone has come to creating an entirely novel virus is likely an experiment conducted by Ralph Baric and colleagues at the University of North Carolina, the U.S. Food and Drug Administration, and the WIV, among other institutions, in which the researchers used the spike protein of an existing bat coronavirus and spliced it to the backbone of a mouse coronavirus. Baric, who was not available for comment, showed his chimera could infect and replicate in human airways cells in vitro and the lungs of living mice. Then the scientists tried to develop a full-length virus, but that proved “significantly attenuated” both in human airway cells and mice. It would need, according to the study, “further adaptation” to become an effective pathogen. And there’s no evidence at all that that derived virus could spread.
 |
For all of Undark’s coverage of the global Covid-19 pandemic, please visit our extensive coronavirus archive. |
Even less is known about the genetics of coronavirus transmissibility, which remains a black box, even as variant after variant has emerged and spread. Racaniello describes these new, spreading variants as simply “more fit.” But other virologists, including Stephen Goldstein, a researcher at the University of Utah School of Medicine, think that in this case “fitness” implies more effective transmission. “The take-home is that that these new variants are worse because they spread more quickly and to more people,” he says. “It all adds up to greater transmissibility.” Evolution towards increased transmissibility is, indeed, a likely scenario, as more transmissible strains should outcompete less transmissible ones, and that seems to have happened here.
But we still do not thoroughly understand the genetics of viral transmission for SARS-CoV-2, or, for that matter, for any other pathogen.
Some people propose that an accidental release of a natural virus, probably a bat virus, triggered the pandemic — but that scenario is no more likely. First, no one has found a bat virus close enough genetically to be the culprit. The bat virus until recently believed to be most closely related to SARS-CoV-2, RATG13, is 96 percent similar. That doesn’t mean, according to coronavirus expert Weiss, that “one small stretch (4 percent of the genome) is different” and the rest is identical. It is different in small ways all across the genome.
Results posted on the preprint server Research Square in September, which have not yet been peer reviewed, suggest three new viruses identified in bats in Laos are even more closely related to SARS-CoV-2. But even assuming that scientists in Wuhan cultured such viruses in the lab — which they told investigators early this year was not the case — this doesn’t mean that a bat-adapted virus escaped from the WIV could have seeded the Covid pandemic. “A virus is never going to come out of a bat ready to go,” says Racaniello. “It never has.”
Bat-borne viruses, including Hendra, Nipah, Marburg, and rabies, can kill people, but they don’t easily spread from person to person. While, in theory, a bat virus that has the ability to infect people via the ACE-2 receptor might be able to spread from person to person, there is no known record of any bat virus (or any other wild animal virus) having done so. Six cases of SARS-like illnesses among miners cleaning bat feces from a bat cave in Yunnan province have been reported, but there is no indication that these cases (three of which were fatal) spread to anyone else. There are known instances of a bat-adapted disease transmitting among people, but always, as in the case of Nipah virus, through very close contact or exposure to bodily fluids, not via airborne transmission. Lab releases of a wild bat virus would necessarily mean that lab workers had to be infected — many of them, to allow transmissibility to evolve — but despite speculation, there have been no reported Covid infections among lab researchers at the WIV.
Proponents of the lab-release explanation also point to prior accidental releases as evidence that this could have happened in Wuhan. Proponents cite the six times SARS-CoV, a related virus that infected more than 8,000 people and killed 774 in 2002-2003, escaped from research laboratories by infecting scientists who passed it on. Or they bring up Janet Parker, who died of smallpox in 1978 when a British scientist, experimenting with smallpox virus, apparently allowed the virus to move through the ventilation system and infect Parker, who was working in a room just above the laboratory.
The difference between SARS-CoV-2 and these instances is that none of them involve new pathogens. Citing instances of the release of pathogens that have already established transmissibility among humans doesn’t begin to address the question of Covid’s origins. And that remains the fundamental question.
But the actual key to Covid’s origins has been there all along. According to Weiss, SARS-CoV and SARS-CoV-2 are more closely related to each other than either is to other human coronaviruses. But Weiss says both are also closer to related bat viruses than they are to each other. After more than a decade of research, it’s been established that the SARS-CoV originated in a bat and then moved, in the live-animal market of Guangdong in southern China, into intermediate hosts, civets, and likely raccoon dogs. Infecting other animals seems to disentangle, so to speak, a well-adapted bat virus from its original host, making it, for a time, something of a generalist, able to infect a range of species, including humans.
SARS-CoV-2 probably evolved in similar circumstances. It is likely that, again, civets, raccoon dogs, or other species acquired a bat-borne virus and spread it to other animals — and then to people: keepers, customers, passers-by in the 1,000-stall Huanan market, where wild animals of many different species were caged together in crowded, filthy conditions. These live animal markets are essentially disease factories, effective laboratories for the evolution of deadly pathogens. Huanan was soon shut down. No outsiders were permitted to examine it, or test workers for seroprevalence, which is, according to Goldstein, a critically important step.
According to the Chinese Academy of Engineering, as of 2016, the exotic food trade was a $19 billion industry in China, out of $76 billion for the overall wildlife industry. Countless live wild animals are sold for the luxury market each year. A lot of money is involved, and there’s a lot of incentive to keep quiet. Though the Huanan market remains shuttered and the wildlife trade for food banned, other markets, selling live animals such as chickens, ducks, and pigs, apparently remain open, and in regions far from the main centers trade in wildlife may continue. Live wild animals are also sold in markets throughout Asia.
Obsession with the lab leak hypothesis, combined with the secrecy and lack of cooperation showed by the Chinese government in helping the world understand the origins of the virus, has taken energy and focus away from an important step that can be taken now to prevent future outbreaks of new viruses. We remain at risk for the evolution of new pathogens, of other pandemics, until all wet markets, worldwide, are shuttered for good.
Wendy Orent, who holds a Ph.D. in anthropology, has been writing about biological weapons and the evolution of infectious disease for 25 years.











Comments are automatically closed one year after article publication. Archived comments are below.
The virus was definitely natural and not lab engineered, but I agree with the the US and international intelligence agencies in suspecting a lab accident, spillover or leak. The BSL suits are a strong point which hasn’t been addressed by the Chinese it the WHO. Also, the covid emergence reported by the US embassy in Beijing early in 2018 and the “mystery illness” reported at the World military games in wuhan in 2019 haven’t been addressed or accounted for.
In the early days of COVID-19 the accepted origin of this coronavirus was that it came from wildlife captured and slaughtered for exotic meat on the Chinese market. This has happened before, like the SARS outbreak of 2002-2004 that began in southern China. For some reason this obvious connection seems to have been forgotten.
We’re not allowed to talk about the origin of COVID-19 because it’s not “politically correct” to do so. That would mean holding a culture accountable for the damage it causes, and that might mean holding our own culture accountable for the damage it causes. Too much work. We’d rather wear masks for the rest of our lives than have an honest conversation.
As you may have noticed, COVID-19 is not going away. And even if it does, there is every reason to expect it to happen again, and again. Have the “wet markets” been shut down? Have people in China stopped eating exotic meat? There is essentially zero reporting on the source of this pandemic. This is a glaring and lethal omission. This is not about blame; this is about accountability.
You can either be politically correct or you can tell the truth. If we actually want to solve our problems I suggest we do the latter.
Wendy,
Thank you for your opinion. However I need clarification; you state that ” To make a human-to-human transmissible disease, you need human beings, a lot of human beings, to be exposed to a pathogen. And you need the repeated action of natural selection on the pathogen spreading among those human beings.”.
So where are the results of lab tests identifying these “interim” infections that would define the evolution of the virus? Also, if your theory of choice, (which I believe is the so called “zoonotic” pathway) is true, then where are the host animals for these interim versions of the virus? If I’m not mistaken all past virus outbreaks that followed this path have resulted in the “host” animals being identified very quickly. Why not this time? How did it get from bats to humans without any identified pathway?
Please enlighten me.
Fundamentally, your points:
1. No close relative of the virus has been found, which would be the case if it was a known virus tinkered with by people in a lab to gain a specific function.
2. No human-engineered virus has ever before been found to become predictably more virulent. It gains one benefit by losing another, unpredictably.
3. Disease vectors like wet markets need to be cleaned up (absolutely!)
4. Mass transmission between many animals is much more effective and quicker to create these pathogens than randomly editing RNA in a lab.
I’ll add a major point from others:
1. The massive improvement in virulence between the original Wuhan and Delta shows the original wasn’t engineered very fit for humans in the first place. Natural transmission makes them stronger, fast.
And further proves your evidence that viruses gain human-human transmission slowly.
Thanks for your good comments – but you appear to be confusing “virulence” (deadliness) with “transmissibility (ability to spread effectively from person to person – or, in animals, host to host).” There is NO “improvement” in virulence between the original Wuhan strain and Delta. (We would expect a DECREASE in virulence based on conventional evolutionary theory, not an increase – that hasn’t happened almost certainly because this stealth pathogen spreads from the apparently uninfected.) It’s the transmissibility and general viral fitness that keeps increasing, not the virulence.
“There is something fundamentally wrong with all lab leak arguments” that SARS2 is human adapted?
No.
Please look up the Mojiang Miner Passage theory
I should write a dissenting article to your opinion piece. It will be titled ‘Why I Still Believe Covid-19 Originated in a Lab’… the Wuhan lab to be exact.
First rule of investigation….follow the smoking gun which is usually the closest & most likely culprit. In this case it would obviously be the Wuhan Institute that was investigating ‘gain-of-function’ into bat coronaviruses. Next look at the denied access to the lab by Chinese authorities until all linking evidence and personnel could be scrubbed from the system. Look at the cursory investigation performed by the WHO that took everything that the Chinese officials spouted at face value.
Next, look at all of the deception put forth by Anthony Fauci of NIH and Peter Draszak of Eco-Health Alliance. It seems as if new disclosures thru FOIA and outside journalistic pressure has caused them to modify their initial denials of funding for gain-of-function and the presence of live bats within the Wuhan Institute. These guys don’t want to be linked to the greatest pandemic in the modern world. They were integral in this fiasco of epic proportions. Why Fauci is still involved issuing edicts on masks and vaccinations behooves me. There is a blatant conflict of interest even commenting on the virus outbreak and proposed remedies.
If none of this raises a red-flag to you, you certainly are not up to task on scientific methodology. I hope that you will eventually open your eyes and see the light.
Such firmly shut eyes seldom open, according to physicist Max Planck: “A new scientific truth does not triumph by convincing its opponents and making them see the light, but rather because its opponents eventually die and a new generation grows up that is familiar with it. . . . An important scientific innovation rarely makes its way by gradually winning over and converting its opponents: it rarely happens that Saul becomes Paul. What does happen is that its opponents gradually die out, and that the growing generation is familiarized with the ideas from the beginning: another instance of the fact that the future lies with the youth.“
Your article proves that my belief that you are a nutcase.
Seems likely that the pandemic started in the Wuhan markets, i.e.,
HUMAN-caused, a direct result of our gross mistreatment and abuse of animals, both wild and domestic. We have very similar markets here in California with animals crammed in cheek-to-jowl with human neighbors, a disaster waiting to happen. Several dozen necropsies on the market frogs and turtles have
documented that ALL are diseased and/or parasitized, though it is ILLEGAL to sell such products. (Cases of E. coli, salmonella & pasturella, all potentially fatal in humans. Worse, the majority of the market frogs carry a dreaded chytrid fungus (Batrachochytrium dendro batidis, or Bd), responsible for the extinctions of some 200 amphibian species worldwide in
recent years. The market animals are routinely bought en mass and illegally released into local waters, where they prey upon and displace the native species, while spreading all sorts of diseases and parasites. This brutal and dangerous trade needs to cease. NOW. The issues are three-fold: environmental protection, public health, and horrendous animal cruelty.
The CA Fish & Game Commission twice voted unanimously to have the Dept. of Fish & Wildlife (DFW) to cease issuing the frog/turtle permits, but were ignored by the Department.
WRITE: Chuck Bonham, Director, Dept. of Fish & Wildlife, 1416 9th Street, Sacramento, CA 95814; email – [email protected]
If the DFW won’t do its job, then legislation is in order.
Interesting point, and I agree.